|
4/15/2023 0 Comments D block elements Platinum, takes on a 6s1 4f14 5d9 configuration and along with gold, these larger elements do seem to be exhibiting some unusual behaviour. There’s also one last additional violator of our Aufbau principle, and that is the element platinum. Oddly though, tungsten beneath molybdenum does not exhibit this kind of behavior because of the difference between the s and d subshell energies for that particular nucleus.Ĭonsequently, so far we’ve got our five exceptions to the Aufbau filling order that we already predicted using filled and half-filled subshells. Not only do chromium and copper exhibit this behavior, so do molybdenum, silver, and gold. Therefore, when we consider the entire d-block, what we find is that most of the exceptions to the Aufbau filling order occur in Group 6 and 11. 
Thus, we have the situation here where chromium and copper, because they have the ability to get half-full or completely full d subshells by snatching one electron away from the 4s, actually fill in an order other than that predicted by the Aufbau principle. It simply steals another one of those 4s electrons away and becomes a 4s1 3d10 in its ground state, another violation of the Aufbau order.įrom here on, we finally reach zinc in which both of these subshells are completely close at 4s2 3d10. Violation of the Aufbau Orderīut there’s also a special energetic benefit associated with a full d subshell, completely full, and this is accessible to a copper atom. And, again, the progression seems to be going along nicely through cobalt and nickel until eventually we reach copper, which, again, the Aufbau order predicts should be a 4s2 3d9. We get to manganese, we again have a 4s2 now 3d5. Instead, the 3d being so close in energy to the 4s is able to steal an electron away, making this a 4s1 3d5 configuration, an apparent violation of the Aufbau order.Īnd then, things get back to normal again for a while. Hence, actually, chromium does not assume the 4s2 3d4 configuration in its ground state. However, a half-filled 3d subshell brings with it an additional sort of energetic benefit. Now at chromium, one might think that it’s 4s2 3d4. This article comes directly from content in the video series Understanding the Periodic Table. We’re going to get additional electrons in the d subshell as we continue stepping across this row until we reach chromium. As one can imagine, this process is going to continue. Moving on, scandium now has a full 4s and also needs to put one more electron in, so it’s going to add that electron to the 3d, giving us 4s2 3d1. As we step again to calcium, we still have space in the 4s subshell, therefore, calcium gets a 4s2 electron configuration. Once we start filling them, potassium, which has a single electron above the argon core, is going to go into the lower energy subshell into the 4s1. What happens is that we progressively add one additional electron for each step over on the periodic table and thus, all of them will end up having an argon core. Let’s fill this region using those elements where those particular subshells that are operative, which would be our 4s and our 3d elements potassium through zinc. 
One can notice that the 3d and 4s subshells are very close in energy with the 3d only slightly higher. To begin with, let’s consider the top of the d-block, that those elements that have 3d subshells, that are filling. 
So, let’s put the d-block elements to the test using the first of these two qualifications-the ground-state configuration. Thus, an element that does not itself have an incomplete d sub-shell may nonetheless be considered a transition element, if it gives rise to an ion with that property. 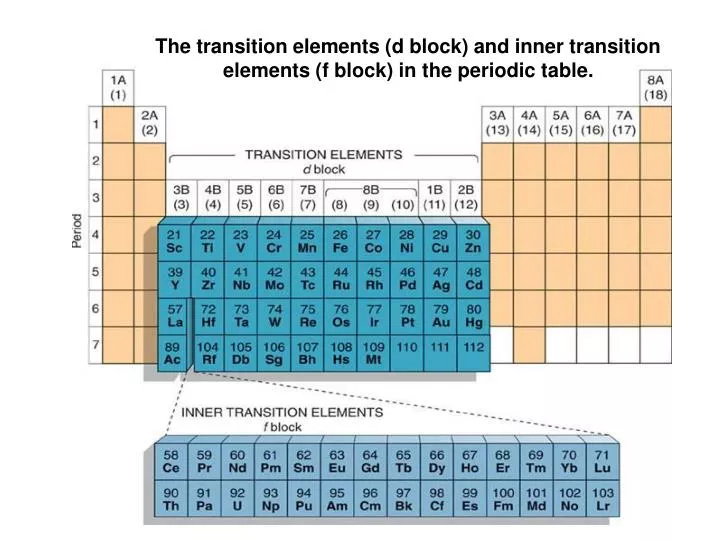
The 1997 IUPAC ‘goldbook’ defined the term ‘transition element’ with a broader, two-pronged definition, as ‘an element…whose atom has an incomplete d sub-shell-without assuming any particular fill order or an element which can give rise to cations with an incomplete d sub-shell. Like Werner’s more modern table, the Aufbau order’s prediction about the fourth-period elements, was an amazing bit of insight years before Schrodinger confirmed the existence and structure of subshells and orbitals.īut as our understanding of atomic structure has continued to evolve, the definition of ‘transition element’ has also changed slightly. Bury proposed that certain properties of elements in what he called the ‘long periods’ could be better explained if elements did, indeed, start to place electrons in a higher energy level, then ‘transition’ back to filling in an interior shell with even more electronsīury suggested that this was exactly what was happening in elements from scandium to zinc, and he was correct! Transition Element (Image: Darren.dolan/Public domain)īut, English chemist, Charles Bury, published a paper challenging Langmuir’s postulate in 1921. Irving Langmuir published a famous paper entitled, The arrangement of electrons in atoms and molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
